 With fewer 90° LP–BP repulsions, we can predict that the structure with the lone pair of electrons in the equatorial position is more stable than the one with the lone pair in the axial position. The central atom is not counted.A linear arrangement means that. The VSEPR theory describes five main shapes of simple molecules: linear, trigonal planar, tetrahedral, trigonal bipyramidal, and octahedral. Observable geometries at an atom are a relationship describing the relative geometric positions of other atoms bonded to that central atom. If we place it in the axial position, we have two 90° LP–BP repulsions at 90°. Why isnt the observable geometry for 'X' linear instead of none You are confusing observable geometry at an atom with molecular geometry. However, because the axial and equatorial positions are not chemically equivalent, where do we place the lone pair? If we place the lone pair in the equatorial position, we have three LP–BP repulsions at 90°. The hybridization of boron in BF 3 is sp 2. The primary difference is that with molecular geometry we include only the.  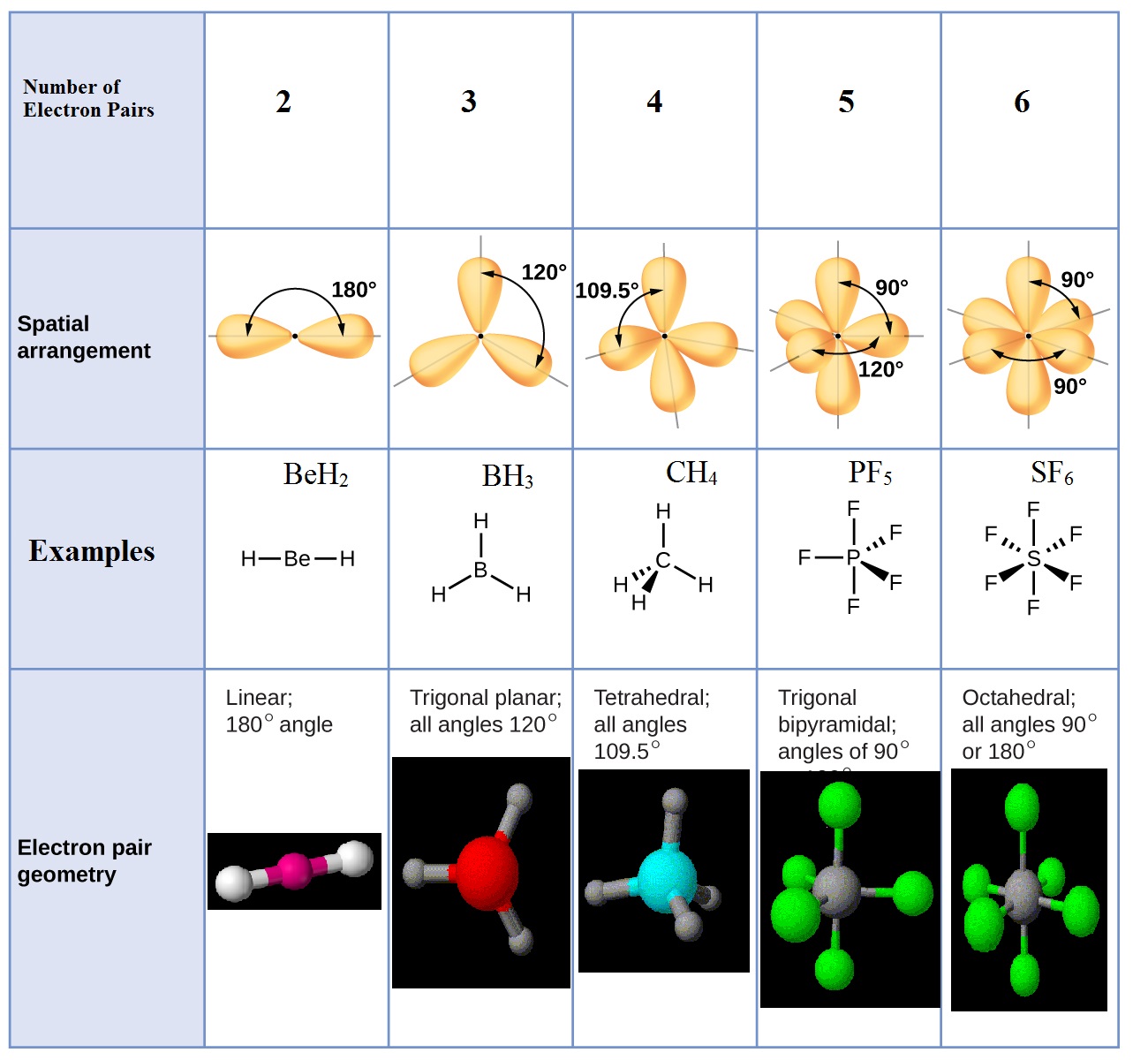
In the BF 3 Lewis dot structure, a total of 9 lone pairs and 3 bond pairs are present. An explanation of the difference between molecular geometry and electron geometry. We designate SF 4 as AX 4E it has a total of five electron pairs. The electron geometry of BF 3 is also Trigonal planar, as its central atom, is surrounded by the 3 regions of electron density. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
